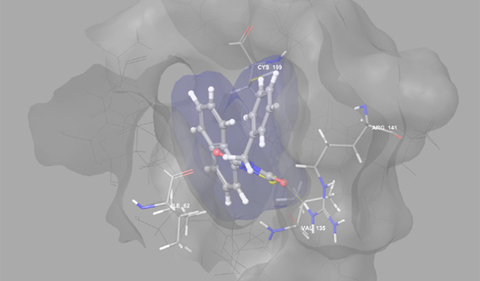
Model of Tideglusib bound to a kinase (GSK3) associated with medical disorders such as Alzheimer’s disease and cancer (image courtesy of J. V. Hines).
By Amanda Biederman
NQPI editorial intern
Douglas Goetz, Chemical and Biomolecular Engineering Professor, Molecular and Cellular Biology Faculty and Nanoscale & Quantum Phenomena Institute member, and Ohio University colleagues were recently awarded a grant from the National Institutes of Health to characterize an inhibitor of glycogen synthase kinase 3 (GSK3), an enzyme implicated in many disorders including diabetes, Alzheimer’s disease and cancer.
The work is a collaboration with Dr. Kelly McCall, OHIO Heritage College of Osteopathic Medicine Professor, and Chemistry & Biochemistry Professors Dr. Stephen Bergmeier and Dr. Jennifer Hines. An unexpected finding during a screening study involving this group and the late Leonard Kohn, a former HCOM member, turned attention toward further investigation of inhibitors, Goetz said.
“We were working on developing a novel therapeutic starting from the drug methimazole commonly used to treat thyroid disease,” Goetz said. “We screened a series of compounds made in (Bergmeier’s) lab, and … one only hit GSK3 alpha and beta.”
A GSK3 inhibitor called Tideglusib was previously designed by another group and tested in clinical trials as a therapeutic for Alzheimer’s disease. However, Tideglusib did not produce a clinical benefit. Goetz said that their inhibitor is both more specific and more potent than Tideglusib.
The group does not yet fully understand the mechanism that accounts for this specificity. They have synthesized structurally similar molecules that also function as GSK3 inhibitors. The team will work to determine the basis of their unique inhibitory function. Hines is exploring their interactions with GSK3 using molecular modeling (see figure). Additionally, the team will introduce their molecules into human macrophage cells to study inhibition in a cellular system.
Broadly, the team is working to determine what makes these small, relatively simple compounds such effective inhibitors. These studies will potentially yield critical data for designing novel therapeutics for GSK3-mediated diseases.



















Comments